Adaptive Bio FDA Panel recommendation for MRD in Multiple Myeloma
Good news for the otherwise battered ADPT stock
The US Food and Drug Administration panel unanimously approved minimal residual disease (MRD) as an accelerated approval endpoint in multiple myeloma drug trials.
Seattle-based Adaptive offers the FDA-cleared ClonoSeq assay for MRD testing in hematologic cancers, including multiple myeloma. The vote from FDA's Center for Drug Evaluation and Research Oncologic Drugs Advisory Committee became public Friday afternoon.
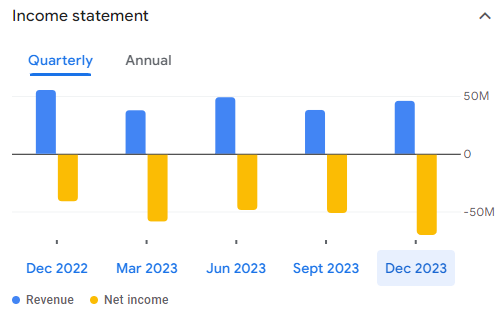
We’ve discussed Adaptive Bio in the past: the company’s ClonoSeq assay is used to characterize the immune cell component of human blood for diagnostic purposes. The company has struggled to show the financial validity of their model, losing a bit more money that they make on revenue on a quarterly basis, which is somewhere around $50M per quarter.
"Many of the 12 voters called today's committee [meeting] a 'Herculean day' for multiple myeloma, and we would argue that today marked a Herculean day also for MRD as an important category of diagnostic testing not only for clinical use, but importantly, for biopharma use in clinical trials," BTIG Analyst Mark Massaro wrote in a note to investors. "Without a doubt, we think today's events are positive for [Adaptive], which is the market leader of hematological MRD testing."
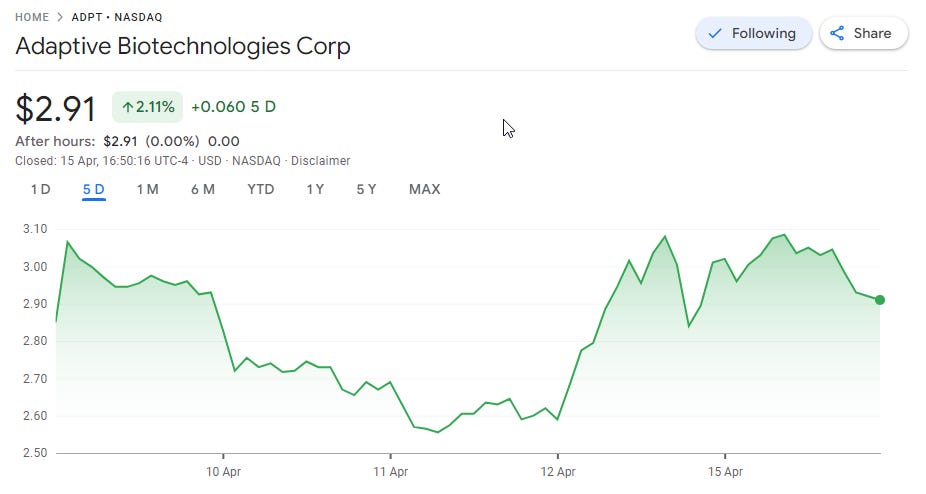
The news of the FDA including ClonoSeq as one of the recommended assays for clinical use but also for biopharma use in clinical trials is a boost to Adaptive Bio ADPT 0.00%↑ , and the stock went up when the news arose.
Still, as the last week or so has been a week of depressed biotech stock valuations, this has only improved the stock for ADPT for the 5-day window. The stock value is still heavily depressed compared to the 2021 biotech boom, where it reached close to $70 per share, now at just below $3 per share.
MRD studies in multiple myeloma use the sequencing-based Adaptive assay or multiparametric flow cytometry, both of which have a sensitivity of 1 in 100,000 cells.
Massaro added that Natera may also ultimately benefit from the decision, predicting that MRD may be used more often as a clinical trial endpoint for solid tumors.
Natera NTRA 0.00%↑ has had a very good run, with many good news coming to them in the last few weeks or months. Although indirectly, this FDA recommendation could also benefit their portfolio, and thus the company stock.
While the panel vote is not the final step in approving MRD as an endpoint, he said he expects the FDA to follow the recommendation.