April 2024 Mass Spec Proteomics
Comparison of the main discovery platforms for Mass Spectrometry Proteomics
Less than a year go, ThermoFisher announced they were bringing a new flagship Mass Spectrometry (MS) instrument to the market: the Orbitrap Astral. Bruker answered by bringing their timsTOF Ultra, and these two instruments now are at the forefront of high-resolution discovery MS Proteomics. Here we describe the capabilities of each instrument and their foundations relative to other commercial offerings in the discovery proteomics landscape.
Both the Orbitrap Astral and timsTOF Ultra offer high scan rate and high spectral resolution which allows each instrument to measure tryptic peptide representations of the protein with high sensitivity, specificity, and robustness. Each instrument is an evolution of the same principals of many other MS instruments on the market. All mass spectrometers can be simplified to building blocks including chromatographic separation, ionization source, ion mobility, ion routing, fragmentation sources, and analyzers.
When it comes to mass spectrometry, the jargon can sometimes feel like deciphering a secret code. However, abbreviations are composed of the components listed above to generalize MS technology. For example, LC-ESI-qTOF reflects a liquid chromatography separation with ESI ionization up front to a TOF MS with peptides separated by quadrupole. A guide to these components is provided at the end of the article. Instrument assembly typically results in a few different categories of mass spectrometer:
Hybrid Mass Spectrometers: An instrument capable of tandem mass spectrometry (MS/MS) for most proteomics experiments. Common configurations include qTOF, q-Orbitrap, and q-LIT.
SciEx 7600, ThermoFisher Exploris 480
Tribrid Mass Spectrometers: A hybrid MS with an additional mass analyzer for more complex experiments, often related to structural studies, but also reflecting the Orbitrap Astral MS.
ThermoFisher Orbitrap Ascend (q-Orbitrap-LIT), ThermoFisher Orbitrap Astral (q-Orbitrap-Astral)
Ion Mobility Enhanced Hybrid Mass Spectrometers: Integration of ion mobility can provide structural and sensitivity-related advantages in analysis relative to non-IM counterparts.
Bruker timsTOF platforms (timsTOF Pro/Pro2, timsTOF HT, timsTOF SCP, timsTOF Ultra, timsTOF Flex)
Triple Quadrupoles (QqQ): These are very fast and attractive for clinical readout on targeted assays, but are not well suited for discovery studies.
ThermoFisher TSQ Quantis, Bruker Evoq Dart TQ, Sciex Triple Quad 7500, Waters TQ XS, Agilent 6495D, Shimadzu 8060NX
While each instrument appears similar in concept, they differ greatly in results, even within manufacturer product lines.
ThermoFisher Orbitrap Astral
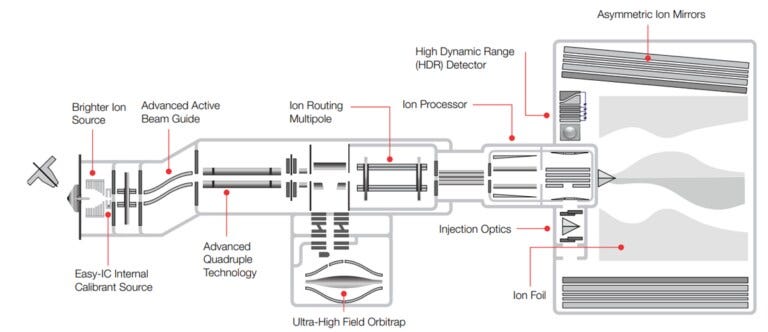
The Orbitrap Astral is an innovative Tribrid instrument that combines an Orbitrap High Resolution with an Astral mass analyzer. The Astral, a TOF analyzer, has been modified to enable higher-resolution data collection compared to most other commercial offerings. In the DIA (Data Independent Acquisition) workflow, the Orbitrap captures intact high dynamic range measurements, while the Astral analyzer performs very fast and sensitive measurements.
Key features of the Orbitrap Astral:
Scan Rates: When paired with its predecessor, the Orbitrap Exploris q-Orbitrap, the instrument achieves scan rates of up to 200 Hz. It captures MS2 spectra at 80,000 resolving power at 200 m/z.
Applications: The Orbitrap Astral has demonstrated its power in various applications, including cell lysate measurements, single-cell proteomics, and plasma proteomics. For instance:
In lysate and tissue samples, it identifies >12,000 protein groups in under an hour (labs of Josh Coons and Jesper Olsen).
Single-cell measurements achieve 6,000 protein group depth in 31-minute gradients (lab of Jesper Olsen).
Human plasma coverage exceeds 10,000 protein groups using Seer-enriched human plasma, with Seer depth per sample reported at over 6,000 protein groups in less than an hour (Seer commercial data).
As part of the ThermoFisher proteomics ecosystem, the Orbitrap Astral is complemented by recommended reagents, materials, and analytics suites. Notable components within the “Velocity” ecosystem include the ThermoFisher Vanquish Neo LC system, uPAC Neo column series, EASY-Spray ESI source, and Proteome Discoverer suite with CHIMERYS. While not mandatory for operating the Astral, these combinations enhance measured sensitivity and robustness within ThermoFisher’s product ecosystem

ThermoFisher Orbitrap Exploris 480
The Exploris 480 preceded the Astral and is subsequently the front half of astral itself. On its own, it is a strong performing instrument that collects data slower and subsequently with less sensitivity than Orbitrap Astral.
Instrument performance highlights include:
480,000 Max Resolution
Up to 40 Hz scan speed (compared to 200 Hz in the Astral)
For all orbitrap analyzers scan rate is inversely related to resolution where 40 Hz is achieved at 15,000 R and only 1.25 Hz is achieved at 480,000 R
m/z 40 to 2000 mass range (40-8000 m/z with pharma option)
>5000:1 dynamic range
Broader range of experiments with multiple scan modes: data-dependent acquisition (DDA), data independent acquisition (DIA), BoxCar DIA and DDA experiments, tSIM, and targeted MS/MS
In Exploris instrument family with Exploris 240 (slightly slower orbitrap), and Exploris 120 (slightly slower orbitrap with software locks)
ThermoFisher Orbitrap Ascend Tribrid
Specs:
Tribrid architecture — includes quadrupole mass filter, linear ion trap and Orbitrap mass analyzers
Ultrahigh resolving power up to 500,000 FWHM
Full parallelization of MS and MSn analyses
Synchronous Precursor Selection (SPS) for MS and MSn experiments significantly increases the number of peptides and proteins identified and improves quantitative accuracy when using isobaric mass tags
Flexibility of fragmentation — CID, HCD, ETD and EThcD available at any stage of MSn
Bruker timsTOF Ultra

In the ever-evolving landscape of mass spectrometry, the Bruker timsTOF platform has emerged as a formidable contender, strategically positioned to rival ThermoFisher’s Q-Exactive and Exploris instrument lines. Over recent years, the timsTOF system has secured a substantial share of the proteomics market, making waves with its innovative features and competitive pricing. Notable elements of the Bruker timsTOF family are as follows:
High Scan Rate and Sensitivity: If the ThermoFisher Orbitrap Astral was a response to anything, it was undoubtedly the Bruker timsTOF. The timsTOF Ultra, in particular, boasts an impressive 300 Hz scan rate and an estimated 40,000 resolving power. It also incorporates low-resolution ion mobility separation via the TIMS IM device.
Market Share: Public datasets indicate that approximately 8% of proteomics datasets (credit: Chris Ashwood) utilize Bruker MS instruments as of 2023, while a dominant 85% opt for ThermoFisher systems. Despite the apparent gap, speculation suggests that the margin between Bruker and ThermoFisher might be narrower than meets the eye.
Three Categories of timsTOF:
Standard: Designed to meet the needs of most users, the standard timsTOF model offers LC-ESI-MS/MS capabilities for analyzing biofluids, tissues, and cell lysates. The flagship of this line is the timsTOF HT, which elevates scan rates and sensitivity to unprecedented levels, surpassing its predecessors—the timsTOF pro2 and timsTOF pro.
Imaging: Tissue imaging has revolutionized the mass spectrometry field, unraveling intricate interactions among lipids, metabolites, and peptides within serial tissue sections. The timsTOF Flex facilitates this exploration by integrating an on-board MALDI source. Think of it as a molecular microscope with a timsTOF lens.
Single Cell: The rise of single-cell proteomics has been nothing short of seismic. Researchers are captivated by the promise of dissecting cell heterogeneity. Enter the timsTOF SCP and the timsTOF Ultra. The SCP, even more sensitive to low-abundance ions than the Pro2, paved the way for the Ultra—a “best hits remix” of SCP and HT capabilities.
Published data is limited on the Bruker timsTOF ultra, but emerging data suggests 6,000 protein groups are measurable in single cells matching that of Astral (lab of Karl Mechtler). Recent research efforts from Bruker commercial teams seem to include immunopeptidomics, an emerging field of measuring peptides presented by MHC class II proteins under immune response conditions. Stay tuned for future benefits to this end.
As with ThermoFisher, Bruker has a host of components to support acquisition including a NanoElute 2 LC, PepSep nanoLC columns, and their real real-time search unit among other components.
SCIEX
SciEx’s strategic advantage lies in its cost-effectiveness. Researchers seeking robust proteomics solutions often find SciEx instruments to be budget-friendly without compromising performance. This affordability has attracted many laboratories, especially those with limited resources.
While SciEx may not match its competitors in absolute depth of measurement, it compensates with unconventional approaches. Let’s delve into two key innovations:
The SciEx 7600 allows implementation of electron-associated dissociation (EAD). Unlike traditional CID and HCD, EAD allows more straightforward measurement of phosphorylated PTMs. CID and HCD often cleave the phosphate PTM from the peptide, complicating analysis. EAD offers an alternate approach, enabling researchers to explore PTMs with greater ease.
The SciEx 7600 also incorporates the ZenoTrap, a sophisticated feature that enhances signal detection. By minimizing noise floors, SciEx achieves better sensitivity and specificity. Compared to counterparts like ThermoFisher and Bruker MS, SciEx’s ZenoTrap approach provides a competitive edge.
Despite its strengths, SciEx faces challenges. The SciEx 7600, while reliable, has struggled to keep pace with the cutting-edge timsTOF and Astral MS architectures. Researchers seeking unparalleled depth have gravitated toward these alternatives, impacting SciEx’s market share.
SciEx’s recent release—the Echo system for MS—has generated buzz. Acoustic waves propel molecules from sample vials into the mass spectrometer at an impressive throughput of over 50 samples per minute. While the Echo system’s appeal is undeniable, its true potential remains untested in the proteomics discovery space. Researchers eagerly await peer-reviewed data to validate its performance for small molecule and metabolite screening and, hopefully, one day proteomics.
Waters Instruments
Waters Corporation offers a diverse range of MS instruments. While not conventionally used in what could be called “conventional discovery proteomics”, Waters’ platforms excel in structural studies.
The SELECT SERIES MRT achieves groundbreaking TOF resolution using Multi Reflecting TOF (MRT) technology similar to Astral. By reflecting ions multiple times between gridless electrostatic mirrors, it extends the ions’ flight path, maintaining sensitivity while significantly enhancing resolution. The Resolution Enhancement Mode (REM) further extends the flight path, resulting in >300,000 FWHM resolution. Unlike other high-resolution mass analyzers, SELECT SERIES MRT maintains consistent mass resolution over a broad mass range and scan speeds. It provides routine part-per-billion (ppb) mass accuracy, reveals fine isotope structures, and ensures high data quality in both MS and MS/MS modes.
The SYNAPT series, including the SYNAPT XS, bridges high-resolution MS and ion mobility separation. It features StepWave XS and Extended TOF (a reflectron). Ion mobility separation allows researchers to explore complex mixtures based not only on size but also shape and charge.
Agilent Instruments
Discovery proteomics instruments are not a key sector in Agilent’s MS product portfolio. They do offer the 6230B qTOF instrument, but acknowledge it as a bridging high-resolution MS for high-mass measurements over nominal mass instruments. Differentiating itself from most other competitors, the 6230B does achieve 20,000 m/z measurements, enabling measurement of heavy, intact protein complexes. Resolving power sits at 20,000 resolution, substantially lower than that of Astral, timsTOF, or Exploris instruments.
Shimadzu Instruments
Like Agilent, Shimadzu does not seek discovery proteomics as a key element of their MS market share. The key instrument that could access this research space is the 9050 qTOF. The key benefit of this instrument is the rapid polarity switching - alternating between positive and negative charged data collection at a much more rapid rate than competitors.
In principle, each of these instrument relies on the same fundamentals of analytical sciences and ion physics, enhanced by commercial IP. Brief and incomplete descriptions of each component in a MS proteomics system are as follows:
Chromatographic Separation:
This process uses liquid chromatography (LC) to separate peptides based on their hydrophobicity. Gas chromatography (GC) can be used, but is more common for small molecule studies.
Longer separation times decrease throughput, but allow for better depth and more accurate quantification of peptides.
Ionization Source:
In discovery proteomics, electrospray ionization (ESI) is commonly used.
For imaging mass spectrometry, matrix-assisted laser desorption/ionization (MALDI) or Desorption ESI (DESI) might be used.
Ion Mobility:
This step is often complementary to chromatographic separation and is related to molecular 3D cross-section.
Bruker’s trapped ion mobility spectrometry (TIMS) technology is a popular commercial solution.
Ion Routing:
Ions are focused throughout the instrument to prevent signal loss and transmit between components.
Quadrupole (q) technology is commonly used for this purpose.
Fragmentation Sources:
To sequence ions effectively, peptides are broken up using energy.
Collision-induced dissociation (CID) or higher-energy collision-induced dissociation (HCD) are common for proteomics studies.
For intact protein measurements, other technologies like Electron Capture Dissociation (ECD), Higher-Energy ECD (EThcD), and Ultraviolet Photodissociation (UVPD) may be used.
Analyzer:
Measurement of peptides can be done in low resolution or high resolution formats.
High-resolution excels in sensitive measurement.
Orbitrap
Time of Flight (TOF)
Fourier-Transform Ion Cyclotron Resonance (FT-ICR; less common for proteomics)
Astral (a multi reflectron TOF, mrTOF)
Low-resolution can offer cost-effective and/or exceptionally fast measurement of ions.
Linear Ion Trap (LIT)
Ion Trap (IT)
Quadrupole (big Q when analyzer, little q when ion routing)
select TOFs
While much of this article has focused on untargeted, predominantly DIA discovery proteomics, stay tuned for future deep dives on the instrumentation available for targeted proteomic assays, structural protein characterization, and clinical measurements.